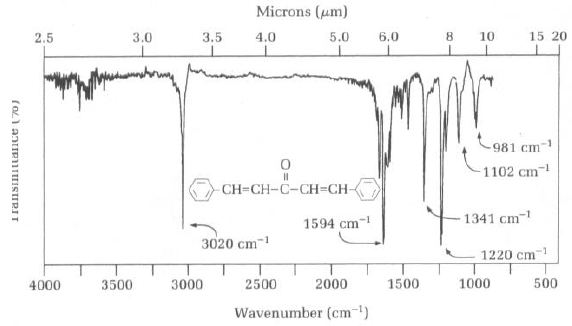
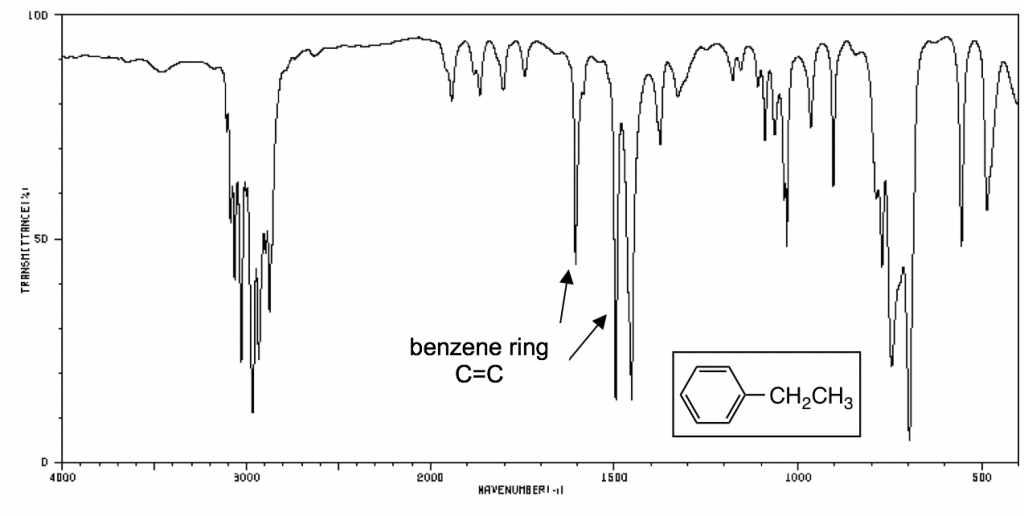
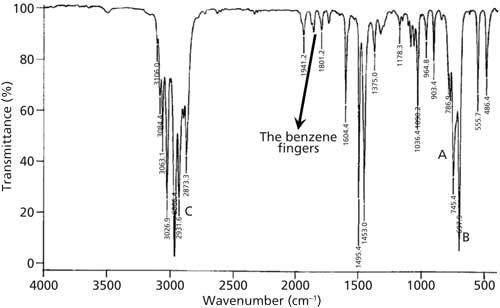
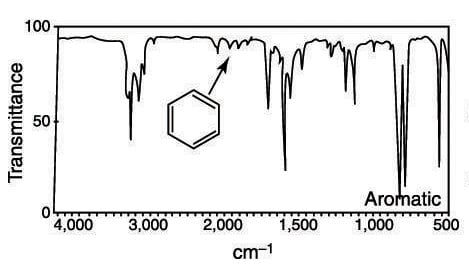
C9H12 infrared spectrum of (1-methylethyl)benzene (cumene) prominent wavenumbers cm-1 detecting ? functional groups present finger print for identification of cumene 2-phenylpropane image diagram doc brown's advanced organic chemistry revision notes

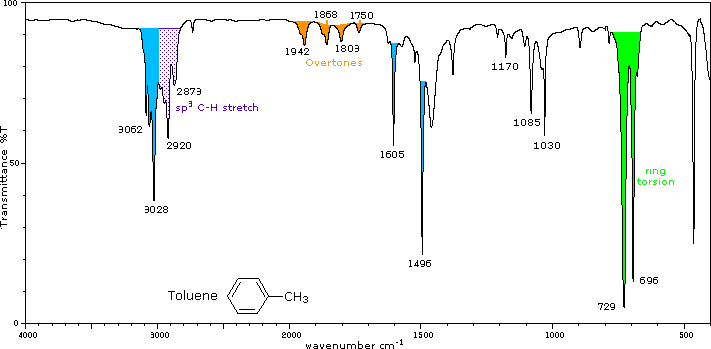
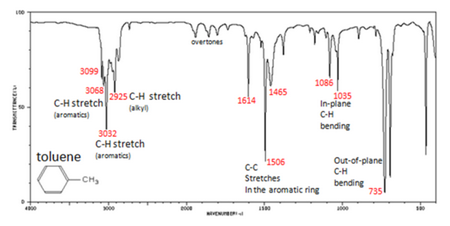
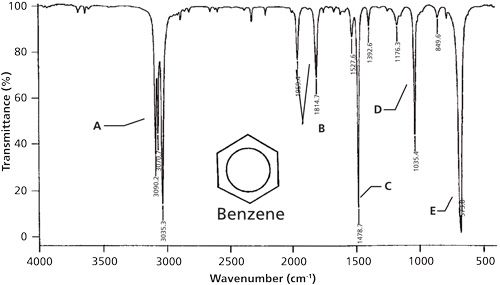
infrared spectrum of methylbenzene toluene prominent wavenumbers cm-1 detecting functional groups present finger print for identification of methylbenzene image diagram doc brown's advanced organic chemistry revision notes

Highly Conjugated, Fused-Ring, Quadrupolar Organic Chromophores with Large Two-Photon Absorption Cross-Sections in the Near-Infrared | The Journal of Physical Chemistry A

Design of All-Fused-Ring Nonfullerene Acceptor for Highly Sensitive Self-Powered Near-Infrared Organic Photodetectors | ACS Materials Letters

Near-Infrared All-Fused-Ring Nonfullerene Acceptors Achieving an Optimal Efficiency-Cost-Stability Balance in Organic Solar Cells | CCS Chemistry

Near-infrared non-fused ring acceptors with light absorption up to 1000 nm for efficient and low-energy loss organic solar cells - ScienceDirect