
Structural and Kinetic Characterization of Diazabicyclooctanes as Dual Inhibitors of Both Serine-β-Lactamases and Penicillin-Binding Proteins | ACS Chemical Biology

The structures of penicillin-binding protein 4 (PBP4) and PBP5 from Enterococci provide structural insights into β-lactam resistance - Journal of Biological Chemistry

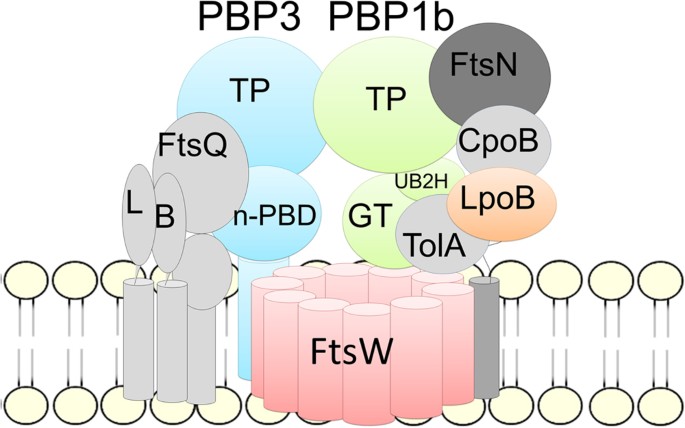
Antibiotics | Free Full-Text | Glycosyltransferases and Transpeptidases/ Penicillin-Binding Proteins: Valuable Targets for New Antibacterials
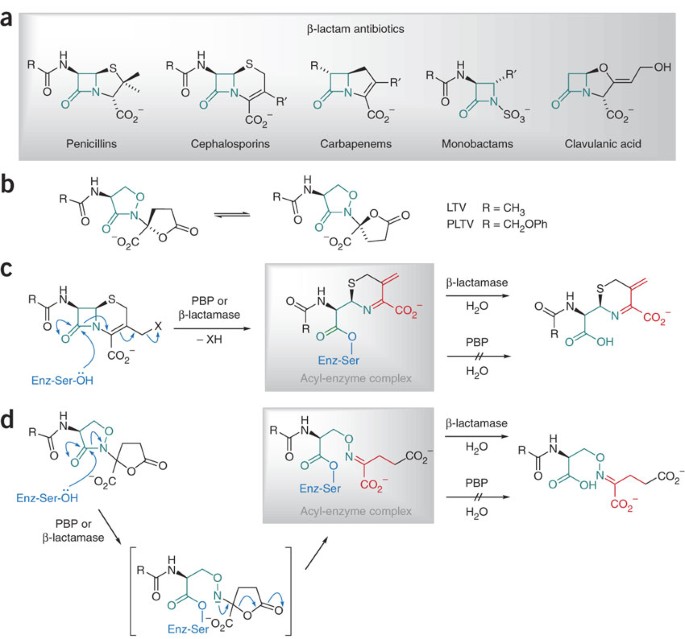
Structural and mechanistic basis of penicillin-binding protein inhibition by lactivicins | Nature Chemical Biology

The Penicillin-Binding Protein PbpP Is a Sensor of β-Lactams and Is Required for Activation of the Extracytoplasmic Function σ Factor σP in Bacillus thuringiensis | mBio
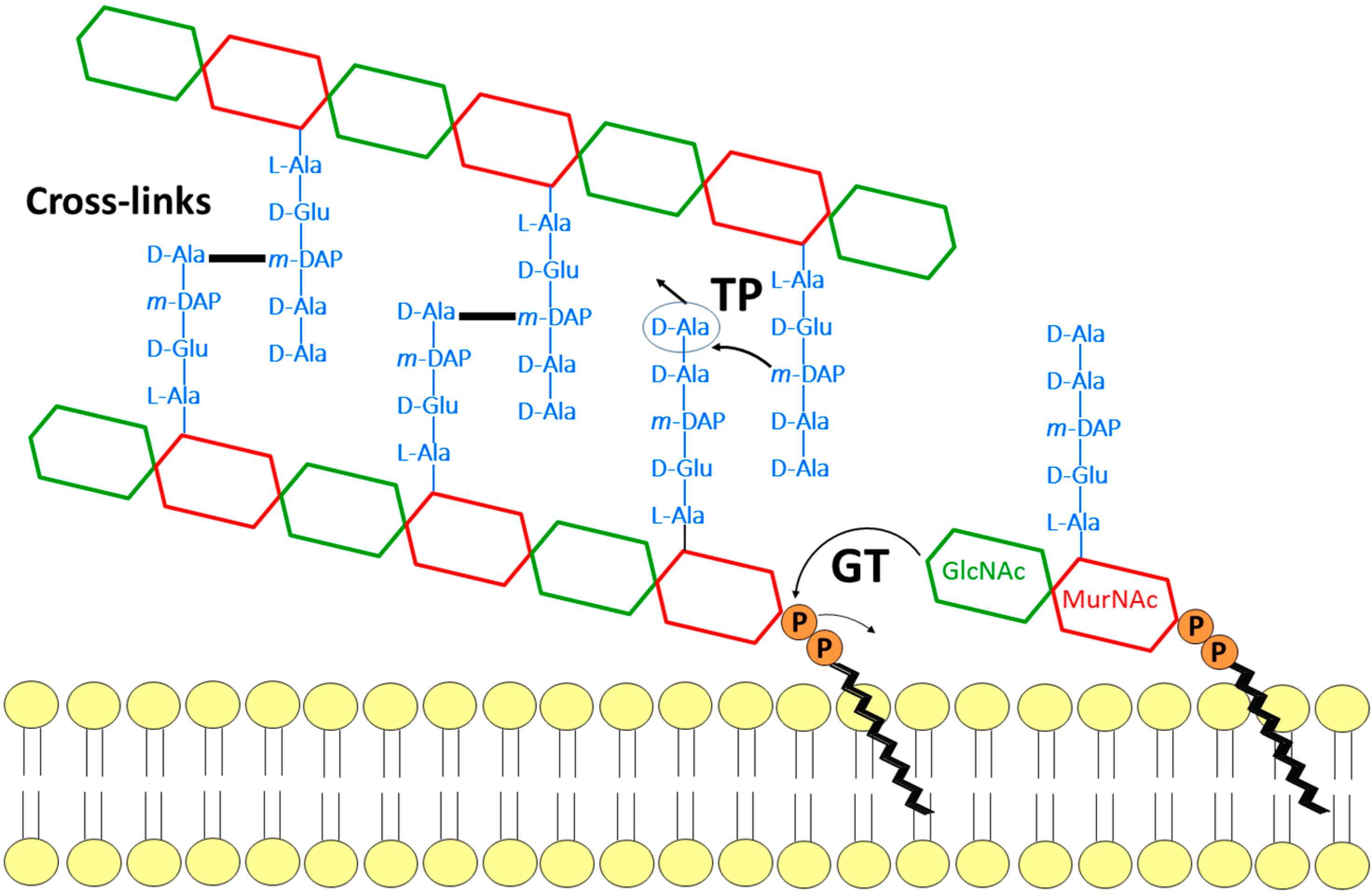
Antibiotics | Free Full-Text | Glycosyltransferases and Transpeptidases/ Penicillin-Binding Proteins: Valuable Targets for New Antibacterials

Osmosis - 💊 Penicillins belong to the pharmacological group of beta-lactam #antibiotics. These all have a beta-lactam ring in their structure, and work by inhibiting cell wall synthesis in bacteria, like Borrelia

An alternative penicillin-binding protein involved in Salmonella relapses following ceftriaxone therapy - eBioMedicine
β-Lactam Resistance Response Triggered by Inactivation of a Nonessential Penicillin-Binding Protein | PLOS Pathogens

Active site restructuring regulates ligand recognition in class A penicillin -binding proteins | PNAS

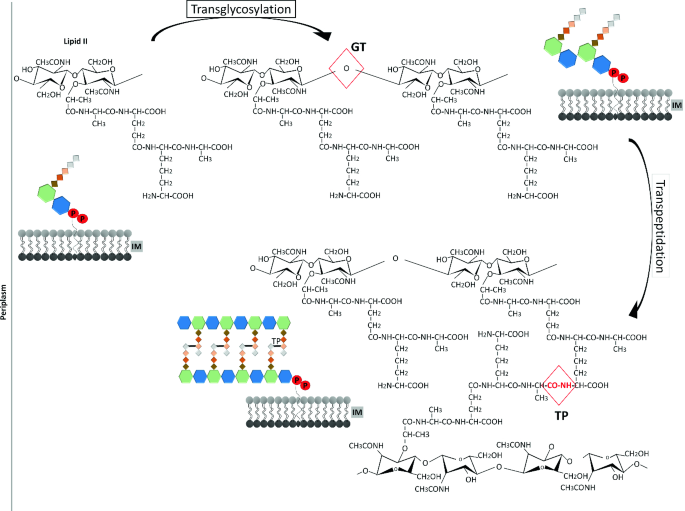

![beta_lactam_working_rough_draft_-_not_ready_for_prime_time [TUSOM | Pharmwiki] beta_lactam_working_rough_draft_-_not_ready_for_prime_time [TUSOM | Pharmwiki]](https://tmedweb.tulane.edu/pharmwiki/lib/exe/fetch.php/betalactammoa.png?w=700&tok=77f359)







