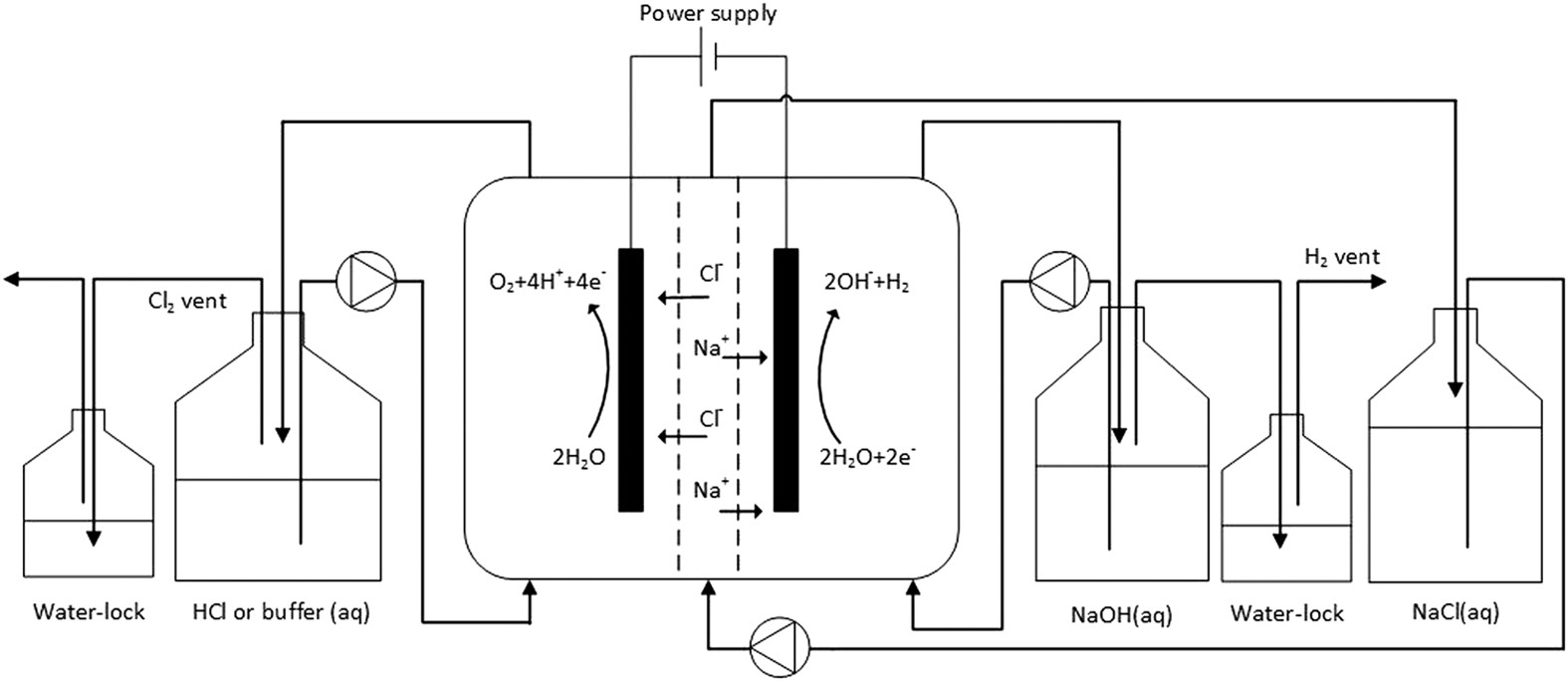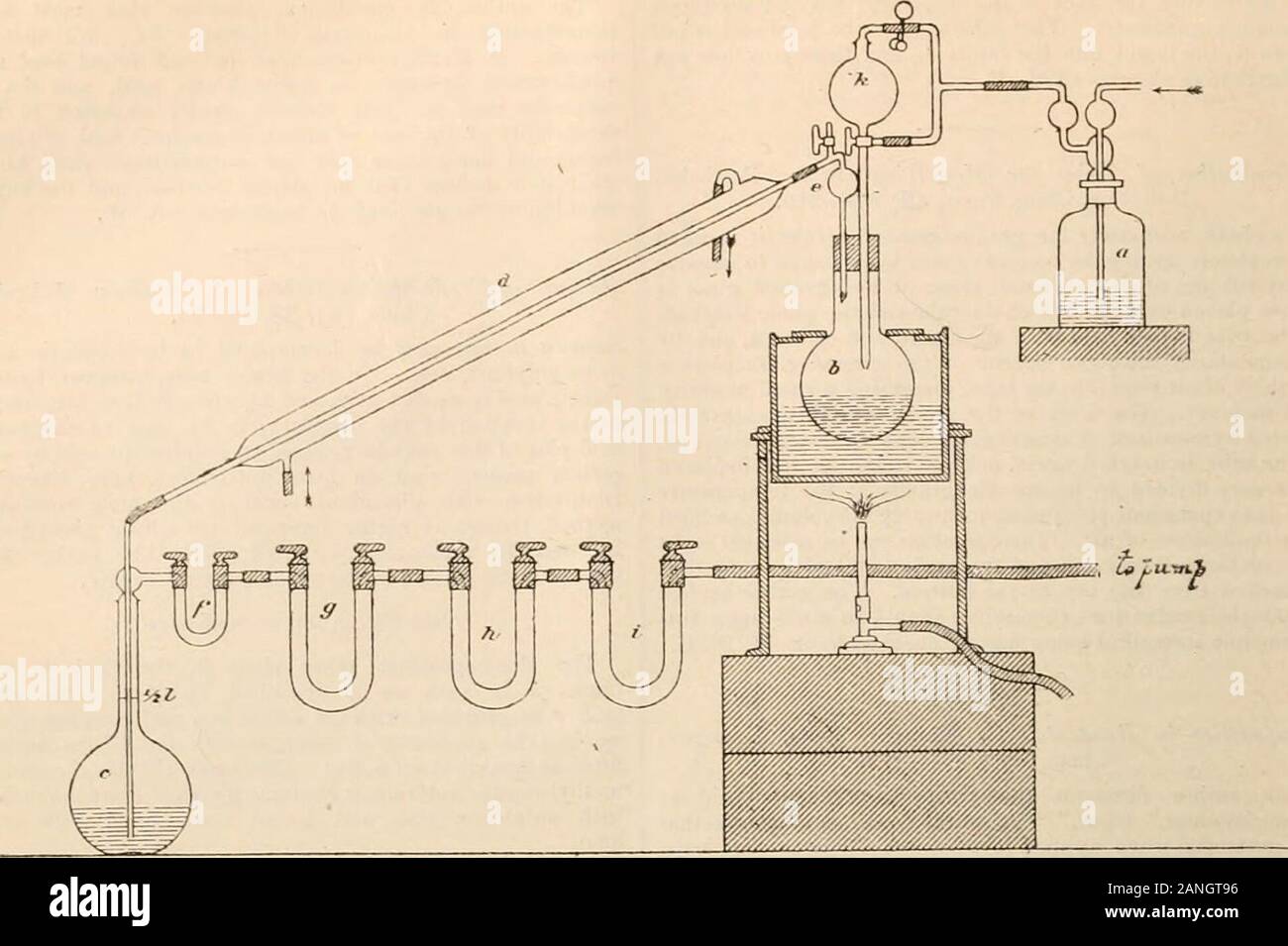
Journal . method. Hedissolves the iron in dilute hydrochloric acid, and passesthe evolved gases by means of ,a stream of hydi-ogenthrough dilute hydrogen peroxide (2-5 per cent.) contain-ing a little caustic soda,

Write the neutralization reaction between Hydrochloric acid HCI and sodium hydroxide NaOH, and write the equation for this process.

Name the products formed in each case when: a. hydrochloric acid reacts with caustic soda b.granulated zinc - Brainly.in

Amazon.com: Hydrochloric Acid - 1 Quart - 32 FL Oz - Muriatic Acid 20 Be - Used in PVC Manufacturing, Regulating PH, Swimming Pools, Salt Purification, Pickling Steel, Leather Processing, Oil : Patio, Lawn & Garden

What is the reason why uric acid is insoluble in hydrochloric acid (HCl) but soluble in sodium hydroxide (NaOH)? - Quora

All I need is hydrogen chloride and caustic soda and I'm about to make some money : r/paydaytheheist