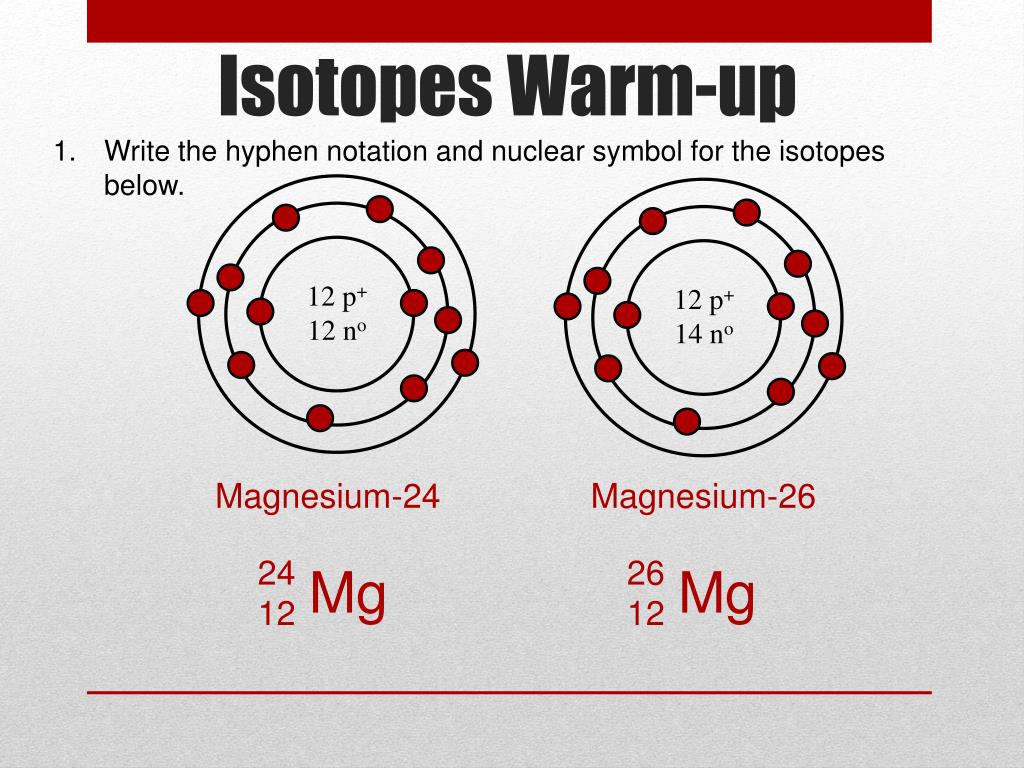
a) 12 Mg and Mg are symbols of two isotopes of magnesium. Compare atoms of these isotopes with respect to: (i) composition of their nuclei. (ii) electronic configuration and valency - CBSE

Magnesium (Z=12) has isotopes that range form Mg-20, "to"Mg-31, "Only" Mg-24, Mg-25,andMg-26 are not radioactive. What mode of radioactive deacy would convert. What mode of radioavtive deacy would convert Mg-20, Mg-21, Mg-22,
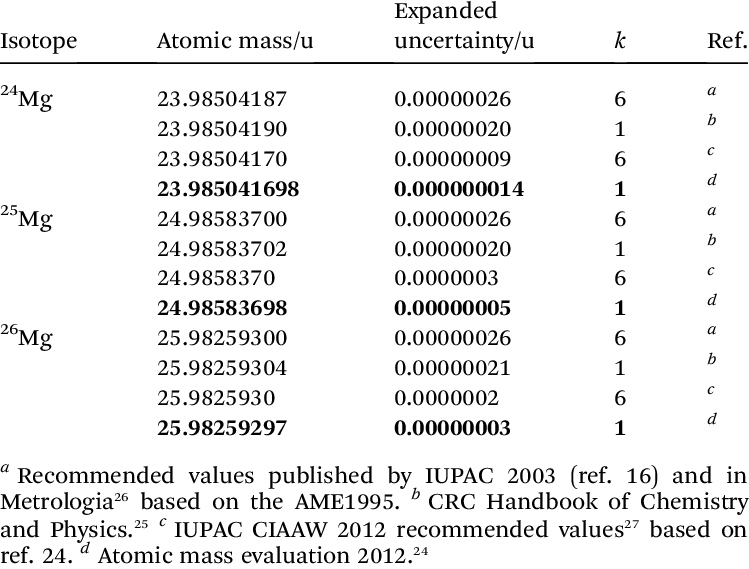
values for the atomic masses of the magnesium isotopes, the constants... | Download Scientific Diagram

Oxygen and magnesium mass-independent isotopic fractionation induced by chemical reactions in plasma | PNAS

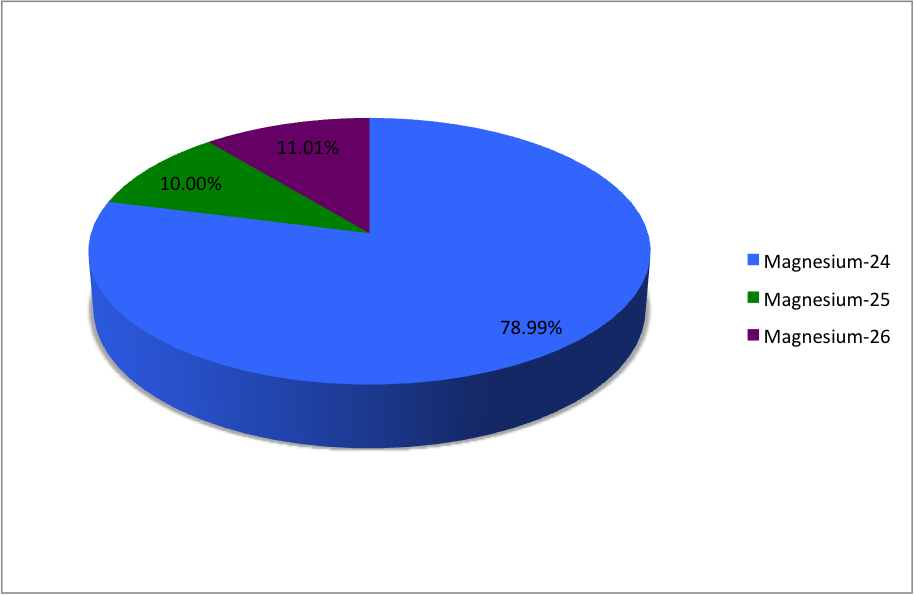
SOLVED:Magnesium has three naturally occurring isotopes: ^24 Mg (23.985 amu) with 78.99% abundance, ^{25} \mathrm{Mg} (24.986 amu) with 10.00% abundance, and a third with 11.01% abundance. Look up the atomic mass of

PDF) Magnesium Stable Isotope Fractionation on a Cellular Level Explored by Cyanobacteria and Black Fungi with Implications for Higher Plants
Magnesium (Mg). Diagramm der atomaren Zusammensetzung und Elektronenkonfiguration eines Atoms von Magnesium-24 (Ordnungszahl: 12), die häufigste Isotop Stockfotografie - Alamy

SOLVED: Calculate the elemental atomic mass of Mg if the naturally occurring isotopes are 24Mg; 25Mg and 26Mg: Their masses and abundances are as follows: Table of magnesium isotopes Atomic Mass Isotopic

Magnesium has two isotopes ^2412Mg and ^2612Mg . According to which isotopes of magnesium have different mass numbers.