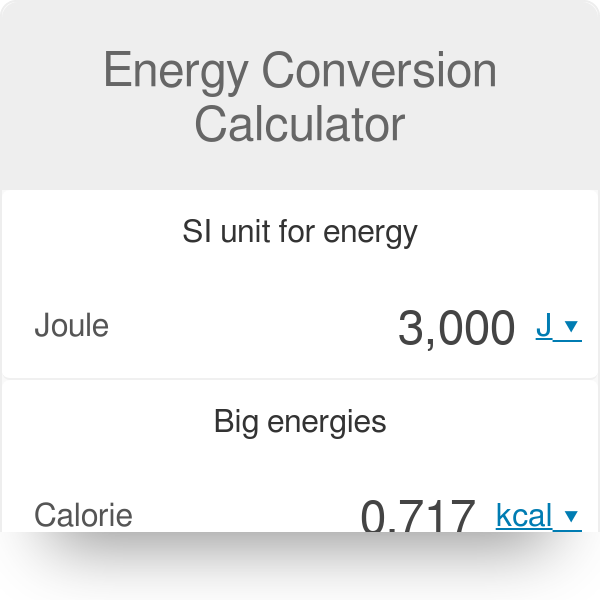
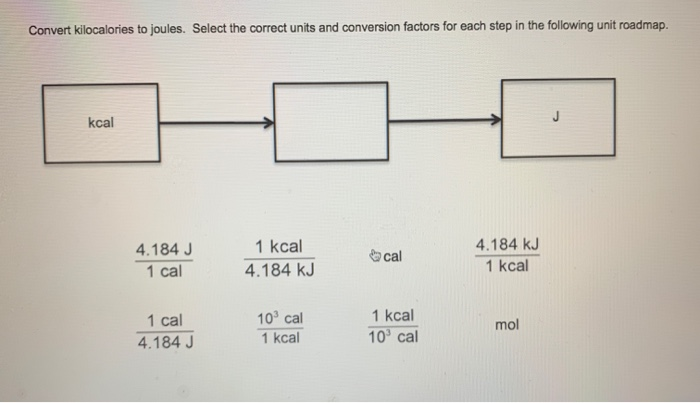
SOLVED:Convert the following numbers of calories or kilocalories into joules and kilojoules (Remember: Kilo means 1000.) a. 75.2 kcal c. 1.41 × 10^3cal b. 75.2 cal d. 1.41 kcal

How To Convert Calories (cal) Into Joules (J) | Factor-Label Method | Dimensional Analysis - YouTube
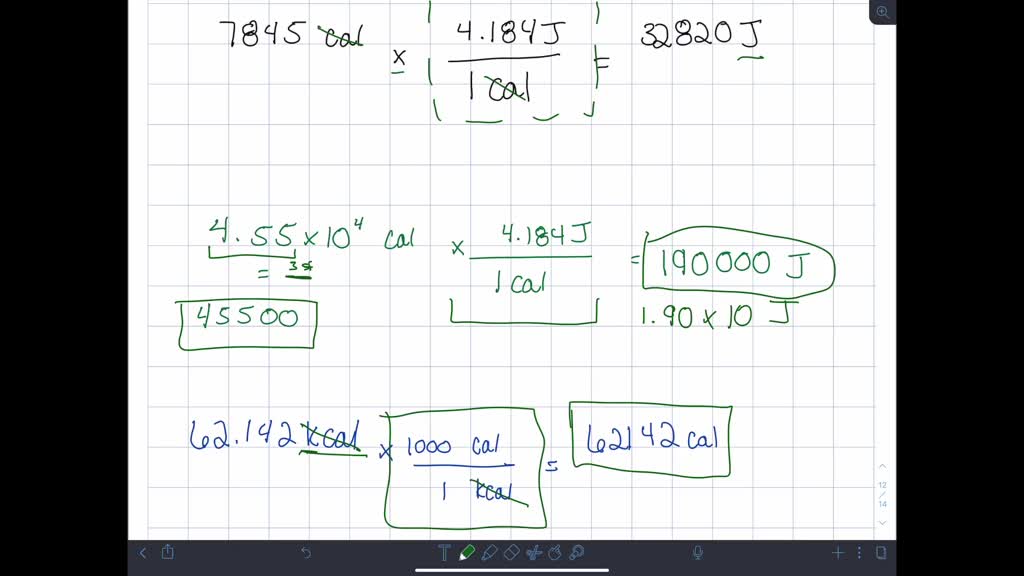
SOLVED:Convert the following numbers of calories or kilocalories into joules or kilojoules. a. 7845 cal b. 4.55 ×10^4 cal c. 62.142 kcal d. 43,024 cal

Question Video: Conversion between Kilocalories and Kilojoules Using the Energy Consumption of the Adult Human Body | Nagwa

The heat Q that must be supplied or removed to change the temperature of a substance of mass m by an amount ∆T is: Q = cm∆T where c is the specific. -