
18.4 g of a mixture of calcium carbonate and magnesium carbonate, on heating, gives 4.0 g of magnesium oxide. The volume of CO2 produced at STP in this process is:
An experiment was carried out to prepare crystals of magnesium sulphate. Excess magnesium powder was - Tutorke

SOLVED: A student is told to make 100 g of magnesium oxide by burning magnesium in excess oxygen: How much magnesium should the student burn? Magnesium reacts with oxygen according to the




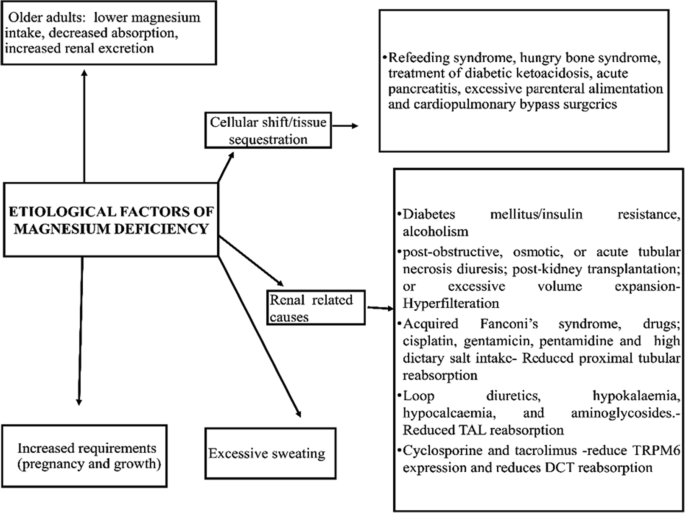

![Etiology of chronic magnesium deficiency [7]. | Download Scientific Diagram Etiology of chronic magnesium deficiency [7]. | Download Scientific Diagram](https://www.researchgate.net/publication/320093091/figure/fig1/AS:1086502096371759@1636053694657/Etiology-of-chronic-magnesium-deficiency-7.jpg)




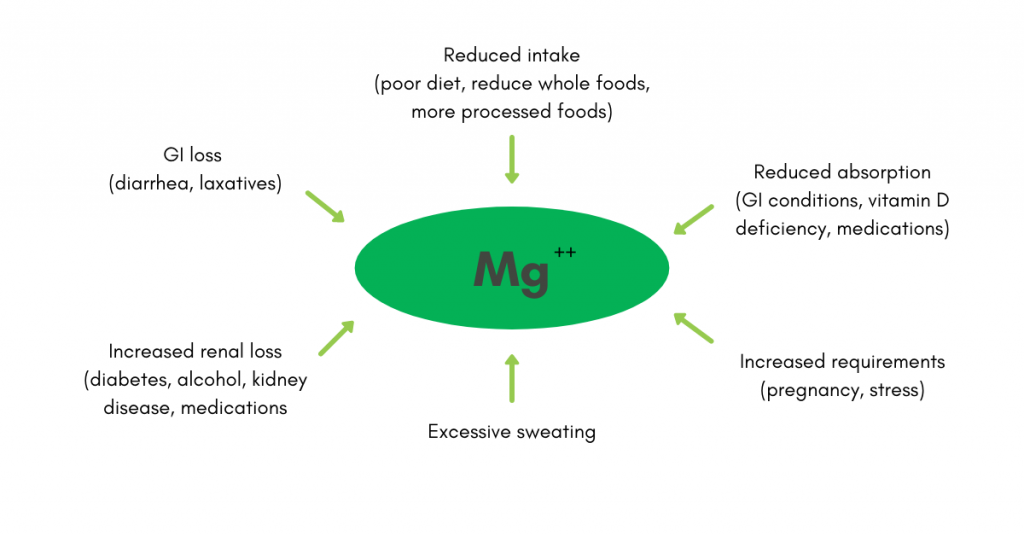
:max_bytes(150000):strip_icc()/magnesium-for-constipation-and-ibsc-1944780_v2-066a0568509b46c497e2fce339b8868e.png)





