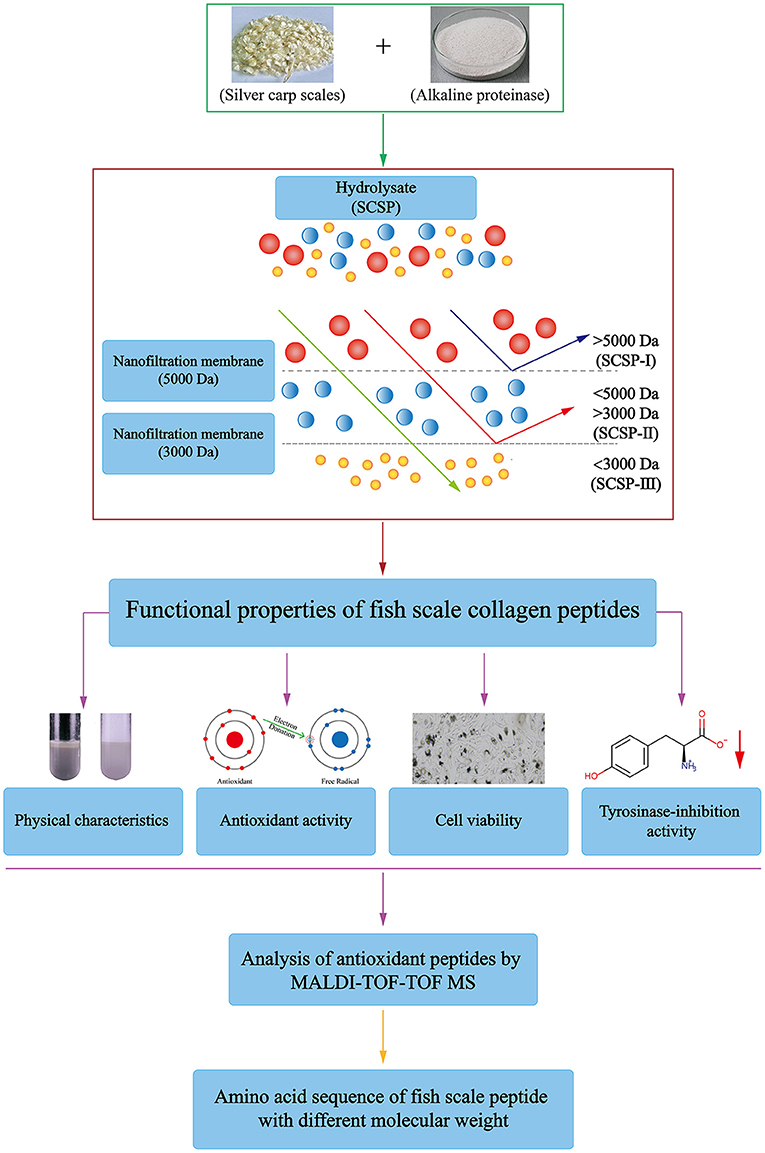
Frontiers | Physicochemical Properties and Biological Activities of Silver Carp Scale Peptide and Its Nanofiltration Fractions

Dalton used the percentages of elements in compounds and the chemical formulas to deduce the relative masses of atoms Unit is the amu(atomic mass. - ppt download
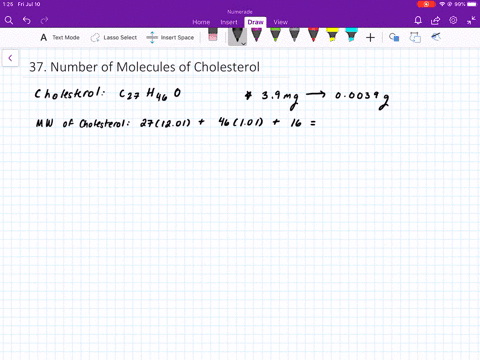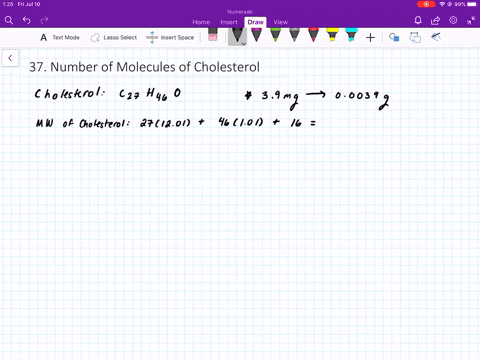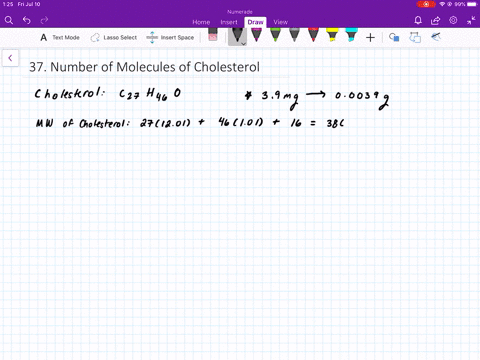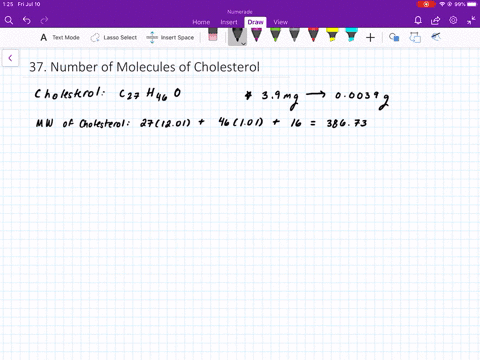
SOLVED:The molecular weight of hemoglobin is about 68,000 amu. What is the mass in grams of a single molecule of hemoglobin?

Dalton used the percentages of elements in compounds and the chemical formulas to deduce the relative masses of atoms Unit is the amu(atomic mass. - ppt download


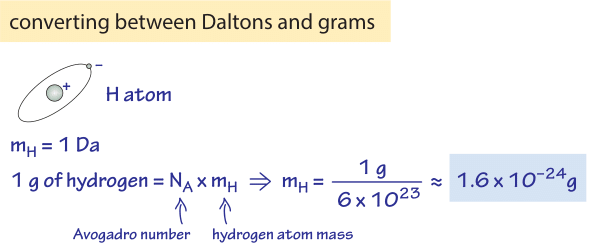








:max_bytes(150000):strip_icc()/atomic-weight-and-atomic-mass-difference-4046144_FINAL_STILL-5940e35000b145ba83fb8e3e40792ba9.png)



